
As I look back on my 20+ years of experience in connected health, I can comfortably say that our team has created connected health innovations that were consistently 5 to 10 years ahead of the market. I offer a few examples here: In the late ‘90s, we were working on videoconferencing as a tool for care delivery. In the early aughts, we started our work on home monitoring for chronic illness; the initial application was for congestive heart failure and our local partner was Partners HealthCare at Home. We took our second opinion program online, launching Partners Online Specialty Consultations (POSC), enabling patients and their physicians around the world to access specialists at Harvard Medical School-affiliated hospitals. Later, around 2005, we saw the need to create a repository for patient-generated data in order to centralize and streamline monitoring programs and pave the way for electronic record integration. Just a few years ago, we accomplished that integration and were considered to be first in the nation to show patient-generated data in the electronic record and the patient portal. In 2010, we demonstrated that follow-up visits for acne could be safely conducted by using an asynchronous online portal. There are now at least five companies offering services in this way.

In each case, when we were doing the work, others were largely disinterested and failed to see the value. Yet, without exception, these innovations are now part of mainstream care delivery or the infrastructure to support it.
I confess to being proud of the team’s innovation record. Watching the process from the inside every day leads to excitement and pleasure. It is fun to come to work in that environment. Of late, we’ve had an upsurge of interest from corporate partners who wish to participate in our brand of innovation. This has led to increased funding and the team is growing.
This all leads me to spend some time reflecting on what is our innovation process. It starts with ideation. Sometimes those are our ideas and sometimes they are ideas brought to us by corporate sponsors. We’ve found that, the earlier we can work with a corporate sponsor — even before product development — the better. Either way, the next phase is iterative.
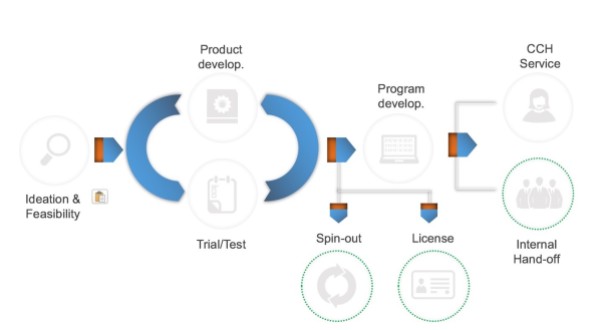
We have a growing user-centered design team that applies a number of tools to get at the heart of how patients will interact with the products we envision. They include interviews, focus groups, ethnography and other capabilities. Once it is determined that we have something people will use, we start the cycle of testing, initially with small groups of patients/users and subsequently with more. At some point, we reach the conclusion that we are ready for large scale validation. That is when we typically initiate a rigorous, IRB-approved clinical trial. We do traditional randomized trials but also case-control studies where we mine our electronic record for a matched control group.
One thing we’ve learned over the years is that a successful clinical trial does not a product make. It is great to see verification of the science behind various tools we develop, but to bring an intervention to the marketplace, another step is required. This is where program development comes in. In this phase, we take the intervention to our Partners HealthCare clinicians (most often primary care practices) and see how it works/evolves in the real world. This phase brings a wealth of worthwhile learning and enables us to move a research program to a product.

From there we can take a number of paths:
- If it is a product we’ve co-developed with a corporate partner, they will have the option to license the intellectual property and commercialize the product. We retain the right to use the product internally at no additional charge.
- If it is our own intellectual property, we can explore creating a company, as we’ve done in the past, or again, seeking a licensing partner.
- Sometimes programs are valuable internally at Partners and not ready for the marketplace externally. In that case, we look for an internal partner to scale the initiative and do what is termed an internal hand off.
- On occasion, we create something that we are confident has market value, but does not have external traction or enough users internally to justify the internal hand off. Hence, we sometimes sunset a program, but at least as often, we choose to operationalize the program internally as we wait for the market to mature.

I’m admittedly proud of our team and the great work they are doing. I’m also curious to learn what innovation looks like in other organizations. Do you have a formal process for developing and vetting products? Are there specific challenges that may be hampering innovation? What are the successes you’ve achieved?
Wanna share this post
- Click to share on Twitter (Opens in new window)
- Click to share on Facebook (Opens in new window)
- Click to share on LinkedIn (Opens in new window)
- Click to share on Tumblr (Opens in new window)
- Click to share on Pocket (Opens in new window)
- Click to share on Reddit (Opens in new window)
- Click to share on Pinterest (Opens in new window)
- Click to share on WhatsApp (Opens in new window)
- Click to share on Telegram (Opens in new window)
- Click to share on Skype (Opens in new window)
